|
| 第18期出刊日:2014.01.10 |
| 化學系周必泰教授研究成果分享 |
| 利用經由水催化之質子轉移互變異構現象探討蛋白質中微水合作用 |
水對蛋白質的動態結構、活性功能、分子之辨識及與酵素之間的相互作用中扮演著關鍵的角色。一直以來,科學家們致力於藉由間接測量的方式,如:分子動力學模擬與/或局部環境極性偵測等等以探索蛋白質內部的水分子。儘管如此,目前仍缺乏直接偵測水分子在生物環境, 尤其是蛋白質裡的方法,藉以從中探索水分子於蛋白質中所扮演之角色。為了解決這個所謂生化水最重要的分子生物課題,我們在這個的研究中突破性的提出了一個以 2,7-二氮吲哚為基礎之色胺酸衍生物——2,7-二氮色胺酸(下稱(2,7-aza)Trp)為生化水分子探針的架構。此分子具有獨特的經由水催化之質子轉移特性。在中性水中,(2,7-aza)Trp 會以兩種質子轉移同分異構物的形式存在:N(2)-H 同分異構物會釋放 380nm 的放光;而 N(1)-H 同分異構物則會因進行經由水催化之激發態質子轉移反應 (excited-state proton transfer, ESPT),產生 340nm 及來自互變異構物 N(7)-H 所釋放鮮豔的綠色放光 (500nm)。這些多重放光性質為我們提供了一個前所未有的機會, 用以偵測蛋白質中的微水合結構以及其在蛋白質活性及功能上所扮演的角色。
我們特別利用了一個重要的蛋白質來展示這個研究的重要性;那就是藉由色胺酸殘基置換成 (2,7-aza)Trp的方式研究目前還是結構未定的人類凝血素合成酶 A2 (Thromboxane A2 Synthase, TXAS)。光譜動力學實驗結果顯示,將位於第 65 残基的色胺酸置換成 (2,7-aza)Trp 後整個抑制了TXAS 之激發態質子轉移效應,這結果顯示了在被置換胺基酸殘基的附近缺乏水分子。反之,置換TXAS 中其餘四個 (2,7-aza)Trp 殘基後, 實驗結果顯示明顯的綠色放光 (500nm),亦即水催化(2,7-aza)Trp質子轉移效應顯著,直接表示了這四個色胺酸殘基皆處於與水分子接觸的環境當中(圖二)。這結果直接印証了同源結晶分析以及分子模擬技術之結果,即位於第 65 残基的色胺酸處於疏水性環境,而其餘四個色胺酸殘基則暴露於與水接觸之蛋白質表面。透過實驗與電腦計算的結合,我們於國際上第一次揭開了水合動力學與蛋白質結構─功能之間的詳細關係。我們相信新發展出的(2,7-aza)Trp分子探針未來將啟動蛋白質中水合現象研究之濫觴。藉由水催化之質子轉移互變異構現象所表現之多重放光性質將引領相關研究深入探討水分子是如何影響蛋白質的折叠、結構與反應活性。
|
| |
Probing water micro-solvation in proteins
by water catalysed proton-transfer tautomerism |
Professor Pi-Tai
Chou(周必泰), Department of Chemistry
In proteins, water plays pivotal roles in their dynamics structures and active functions as well as in phenomena such as molecular recognition and enzyme interactions. Scientists have made tremendous efforts to gain understanding of the water molecules in proteins via indirect measurements such as molecular dynamic simulation and/or probing the polarity of the local environment. However, sensing waters or more specifically biowaters with a more direct manner and hence their possible functionality in protein still lacks. In this contribution, we present a new 2,7-diazaindole based tryptophan analogue (2,7-aza)Trp that shows unique water catalyzed proton transfer properties. In neutral water, (2,7-aza)Trp exists in two proton-transfer isomers: The N(2)-H isomer exhibits a normal 380 nm emission, and the N(1)-H isomer undergoes water catalyzed excited-state proton transfer (ESPT), giving an N(1)-H (340 nm) and a prominent green N(7)-H tautomer (500 nm) emission (Figure 1). The characteristic multiple emissions offer the unprecedented opportunity in sensing the water micro-solvation of proteins.
An exquisite case in point is demonstrated by examining the structurally pending human thromboxane A2 synthase (TXAS) by site-specifically replacing Trp residues with (2,7-aza)Trp. Spectroscopic data mark the lack of ESPT in (2,7-aza)Trp65 of TXAS, implying a lack of water in its proximity. Conversely, as evidenced by the prominent green (500 nm) emission and thereby ESPT, the other four (2,7-aza)Trps are in water-contact environments (Figure 2). The results prove the homologous crystallographic and modeling structure showing that Trp65 is in a hydrophobic environment, whereas the other four Trps are in the probe-exposed site at a protein surface in contact with water. The combination of experiments and a computational framework reveals the intimate relationship between water hydration dynamics and protein structure-function. These results verify that TXAS possesses a peripheral phenylalanine-cluste gating site, which may be crucial in the recognition of ligands in the key early step of allowance to the active heme site. We thus demonstrate for the first time the feasibility of the novel (2,7-aza)Trp, which serves as an ideal probe for sensing water environment in protein without disrupting its native structure.
Because the aqueous micro-solvation phenomena are of particular interest with emphasis on understanding their structures particularly in protein, we believe that our newly developed (2,7-aza)Trp will stimulate considerable future work on characterization of fundamental aqueous hydration phenomena. The characteristic multiple emission manifested by water catalyzed proton transfer tautomerism may lead to future research to probe how water molecules affect the folding, structures and activities of proteins.
|
 |
Figure 1 The proposed schematic diagram proton transfer in water for 2,7-diazaindole and (2,7-aza)Trp in electronic ground and excited states. The barrier and hence potential energy surface are arbitrarily chosen.
|
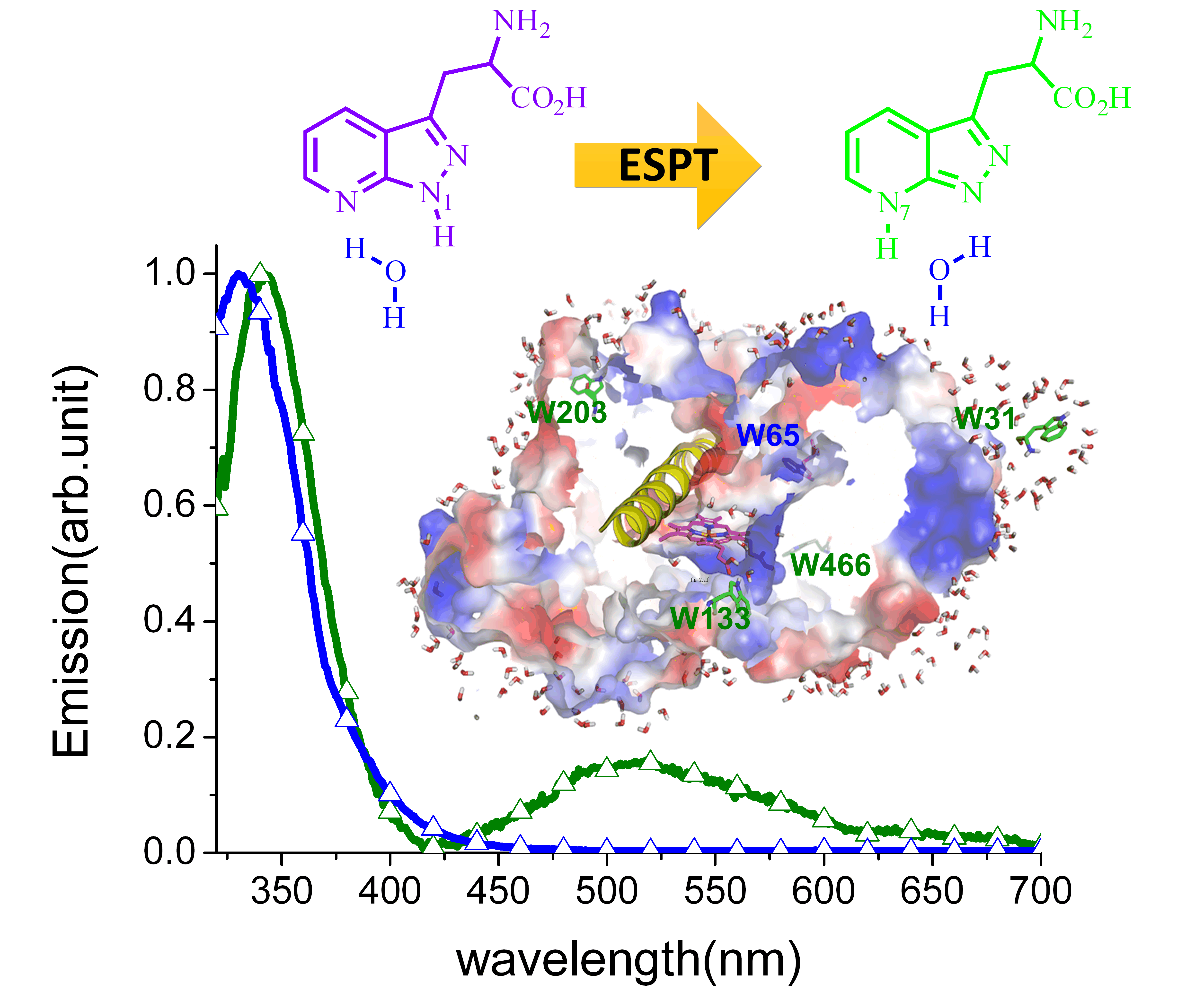 |
Figure 2. As revealed in the emission spectra, replacing Trp31 with (2,7-aza)Trp in human thromboxane A2 synthase (TXAS) gives a prominent green emission band, indicating the occurrence of water catalyzed excited-state proton transfer (ESPT). Conversely, evidenced by the 340 nm emission only, ESPT is prohibited in (2,7-aza)Trp65 of TXAS, implying a lack of water molecules in its proximity. The results prove the homologous crystallographic and modeling TXAS structure, in which Trp65 is in a hydrophobic phenylalanine cluster (Phe-cluster) environment, whereas the other four Trps are in the probe-exposed site at a protein surface in contact with water.
|
| |
 |
| |
|
|



